  
This phenotype correlates with reduced male fertility and an increase in nondisjunction events both in meiosis I and meiosis II. Live cell imaging reveals defects during meiotic divisions after RNAi-mediated knockdown of condensin I-specific mRNAs. This study shows that condensin I-specific subunits localize to meiotic chromatin in both meiosis I and II during Drosophila spermatogenesis. The role of condensin I during male meiosis in Drosophila has been unresolved. However, meiotic divisions in Drosophila males require functional condensin II subunits. In Drosophila melanogaster, faithful mitotic divisions depend on functional condensin I, but not on condensin II. While the different contributions of the two common condensin complexes, condensin I and condensin II, to chromosome morphology and behavior in mitosis have been thoroughly investigated, much less is known about the specific roles of the two complexes during meiotic divisions. The heteropentameric condensin complexes play vital roles in the formation and faithful segregation of mitotic chromosomes in eukaryotes. Condensin I is required for faithful meiosis in Drosophila males. Kleinschnitz, K., Viessmann, N., Jordan, M. Together, these results provide the first causal evidence that proper CT formation can protect the genome from potentially deleterious translocations in the presence of DNA damage. Moreover, Condensin II over-expression is sufficient to drive spatial separation of CTs and attenuate the translocation potential of cells. Using whole-chromosome Oligopaints to identify genomic rearrangements, this study finds that increased contact frequencies between chromosomes due to Condensin II knockdown leads to an increased propensity to form translocations following DNA damage. This question was addressed in Drosophila cells by modulating the level of Condensin II, which regulates CT organization. However, it remains unclear if CT formation itself influences the translocation potential of cells. This is supported by correlations between the frequency of inter-chromosomal contacts and translocation events in myriad systems. 00199-0/attachment/ec086768-c1c4-47ba-bec9-4352334ebd8e/mmc1.jpg)
The formation and spatial arrangement of chromosome territories (CTs) in interphase has been posited to influence the outcome and frequency of genomic translocations. 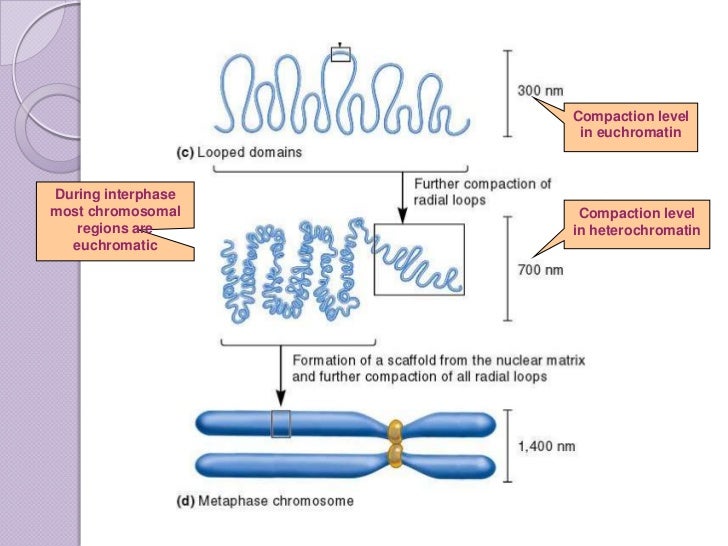
Chromosome territory formation attenuates the translocation potential of cells. This study reports evidence of rapid evolution and recurrent positive selection in seven centromere-associated proteins in species of the Drosophila melanogaster subgroup, and further postulate that positive selection on these proteins could be a result of centromere drive and compensatory changes, with kinetochore proteins competing for optimal spindle attachment. Missense mutations in these proteins often result in improper centromere formation and aberrant chromosome segregation, thus selection for maintained function and coevolution among proteins of the complex is likely strong. Rapid evolution of both Cid and centromeric satellite repeats led to a hypothesis that the apparent compensatory evolution may extend to interacting partners in the Condensin I complex (i.e., SMC2, SMC4, Cap-H, Cap-D2, and Cap-G) and HP1s. Cid, a protein that mediates kinetochore/centromere interactions, displays particularly high amino acid turnover. Rapid evolution of centromeric satellite repeats is thought to cause compensatory amino acid evolution in interacting centromere-associated kinetochore proteins. Widespread positive selection drives differentiation of centromeric proteins in the Drosophila melanogaster subgroup. Genetic map position - 2L:20,058,164.20,060,975 Ĭlassification - Condensin, subunit H, Barren proteinīeck, E. Keywords - mitotic chromosome condensation, promotion of sister chromatid resolution, maintainance of structural integrity of centromeric heterochromatin 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
